L3 Filarial Parasite Chemotaxis Assay
Materials
- Agarose (Molecular Grade)
- 6 cm Petri Dishes
Preparing Chemotaxis Plates
Note: Make chemotaxis plates at least 48 hours in advance of assay. Humidity plays a significant role in plate optimization for this assay.
-
Make 0.8% (w/v) agarose plates. Microwave to dissolve (mix intermittently). Pour ~10 mL of agarose into each 6 cm plate. Leave plates at room temperature with lids cracked for 24 hours.
-
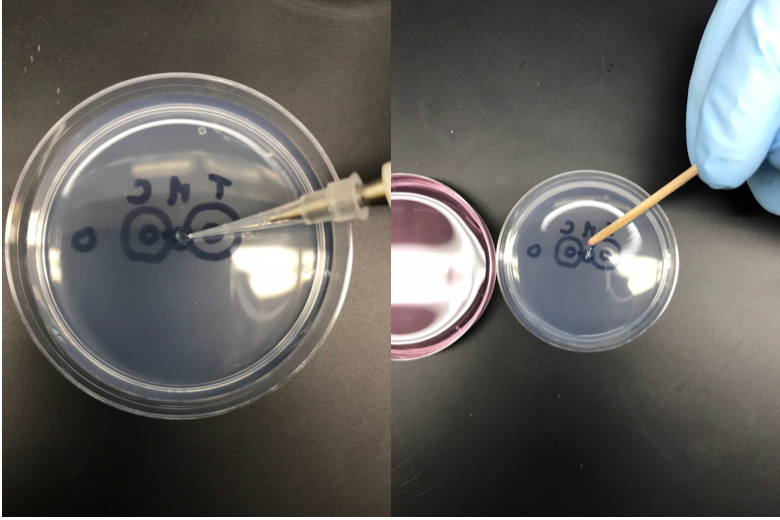
Measure (using stencil) and label the chemotaxis plate as in the image below:
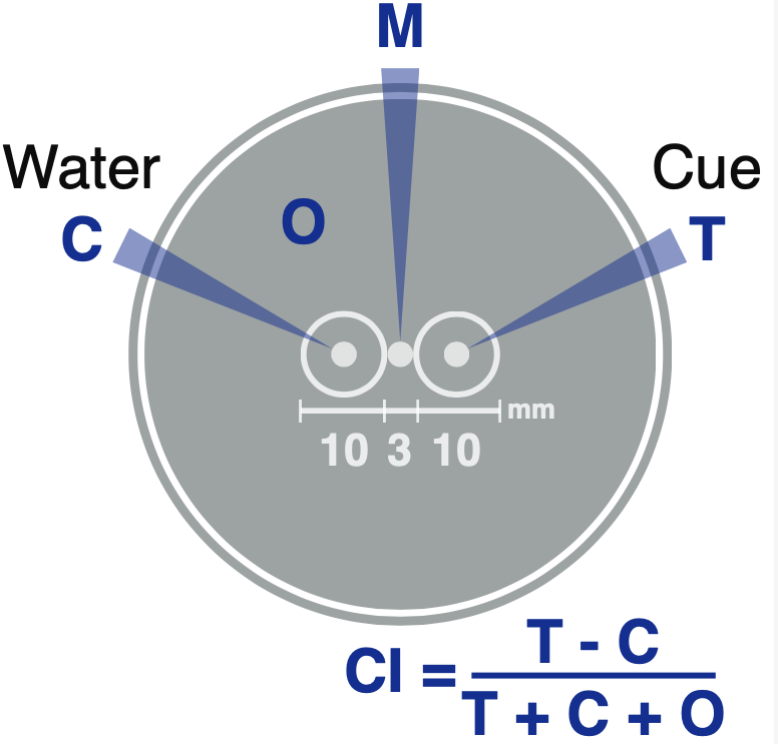
Assay Setup
-
Allow L3/L4 to soak in RPMI+P/S for ~30 min prior to chemotaxis assays to re-sensitize to FBS
-
Place 2 µL of DI H2O in the M-area, and then pick 8-10 L3s/L4s and place in the DI H2O in M-area
-
After L3s are placed, add 3 µl of test compound into the T-zone and 3 µl of DI H2O into the C-zone
-
Place plates in 37°C incubator monitor the L3 migration every 10 min.

-
Once majority of L3 have begun to migrate, count the L3s in each zone/area. This includes counting the L3s that have not left the center.
-
Run each condition in at least triplicates.
-
Store data using template .csv files in the appropriate Box directory and use existing scripts to calculate the chemotaxis index (CI).